Your Trusted Partner In Delivering Software Validation Solutions
Our scalable, flexible, and transparent support helps you focus on your goals, while we ensure software validation becomes an integral part of your workflow.
Validation You Can Trust. Partnership You Can Count On.
GMP Navigator specializes in delivering end-to-end software validation services for regulated industries. We help businesses ensure that their software systems work exactly as intended. Through Computer System Validation (CSV), we make sure your software solution is reliable, secure, and fully compliant with industry standards like FDA 21 CFR Part 11, GAMP 5, ISO, EU Annex 11, and FSMA. Our risk-based , audit-ready approach, helps your business implement and maintain compliant, reliable, and secure software systems.
Our scalable, flexible, and transparent support helps you focus on your goals, while we ensure software validation becomes an integral part of your workflow.
We serve the Life Sciences, Medical Devices, Nutraceutical, and Food & Beverage sectors, delivering tailored validation solutions backed by deep regulatory and ERP software expertise, along with regulated industry domain knowledge. This makes us your perfect partner to ensure that your ERP, QMS, WMS, MES, and other software systems meet regulatory requirements and comply with global standards.


Why Validation Matters?
Validation is not optional — it's critical for delivering safe products, maintaining compliance in regulated environments. It matters because it:
- Ensures systems are compliant, reliable, and secure.
- Confirms software performs as intended and meets the obligatory regulatory requirements.
- Ensures product Quality & patient Safety through consistent, accurate, and traceable results.
- Supports data integrity and trustworthy records.
- Audit Readiness by providing documented evidence to meet inspection requirements.
- Risk based validation approach minimizes chance of non-compliance, recalls, and penalties.
- Improves operational efficiency and enables business continuity.
Services We Offer
Our flexible engagement models are designed for businesses of all sizes.

Complete ERP Software Validation
Complete end-to-end ERP Software Validation

Validation Toolkit
Ready-to-use templates aligned with norms like FDA & GAMP 5

Complete QMS Validation Services
Complete end-to-end QMS validation service

Validation-as-a-Service (VaaS)
Ongoing validation & monitoring for contract-period

Gap Assessments & Training
Identify gaps, train your team, and always stay compliant
Industries We Cater
We bring comprehensive validation expertise to every project. Our priority is to help you meet regulatory requirements seamlessly, without disrupting your operations.


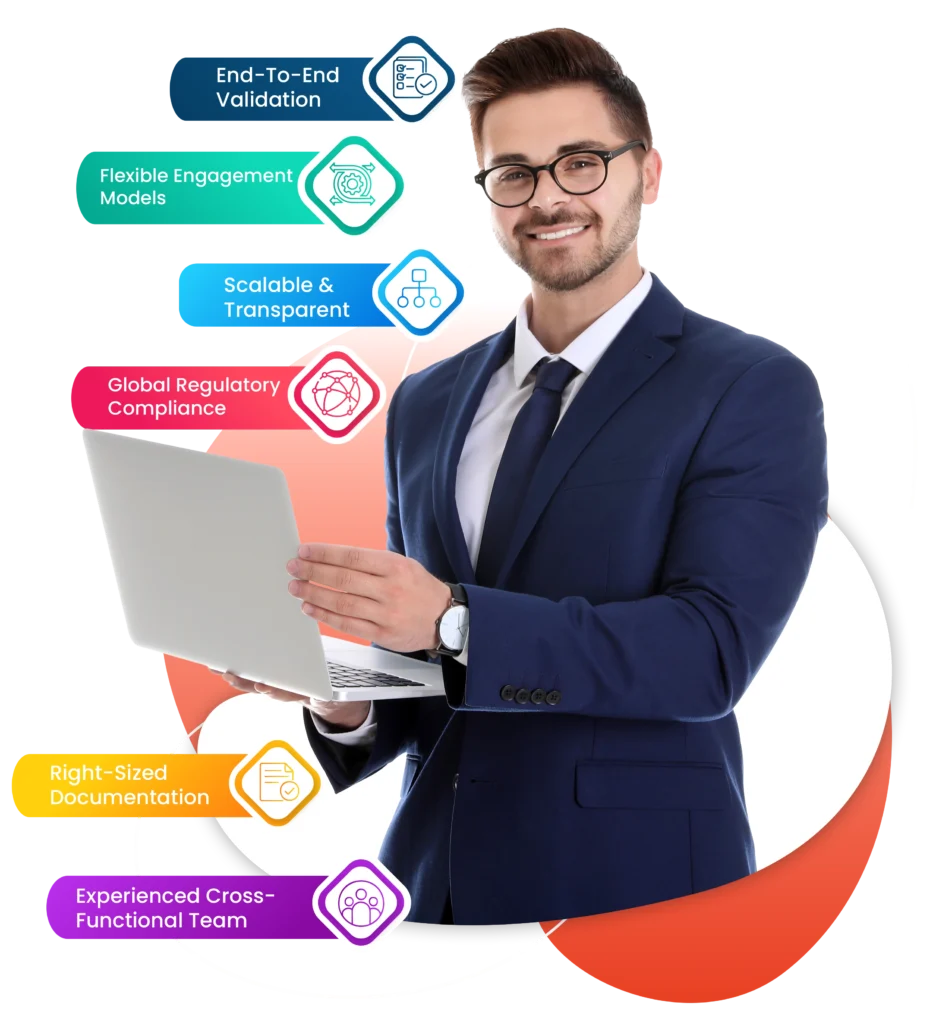
Why Choose GMP Navigator?
Because with GMP Navigator, validation is done right.
- End-to-End Validation - Comprehensive support from User Requirements (URS) to Final Validation Reports.
- Multi-System Expertise - Proven experience with ERP, QMS, WMS, MES, and other critical systems.
- Global Regulatory Compliance - Alignment with FDA, GAMP 5, 21 CFR Part 11, Part 210/211, Part 820, FSMA, and more.
- GAMP 5-Compliant Documentation - Risk-based, audit-ready documentation that meets industry standards.
- Flexible Engagement Models - Choose from Validation Toolkit, Complete Software Validation, Validation-as-a-Service (VaaS), Gap Assessments, and Training.
- Experienced Cross-Functional Team - Deep domain knowledge in regulations, ERP systems, and industry processes.
- Reliable, On-Time Delivery - Projects completed on schedule and within budget, every time.
The Approach We Follow -
GMP Navigator applies a risk-based, GAMP 5-compliant methodology customized to your system and regulatory landscape. Our approach ensures efficient validation, reduced compliance risks, and full alignment with global quality standards.
How We Do It
We support validation for both on-premises systems and Cloud/SaaS Platforms.
System classification and risk assessment as per GAMP 5 guidelines.
- Validation Master Plan (VMP)
- User Requirements Specification (URS)
- Functional Requirements Specification (FRS)
- Risk Assessment
- IQ/OQ/PQ Protocols & Reports
- Traceability Matrix
- Final Summary Report
Validation of system setup, access controls, user roles, audit trails, and e-signatures to ensure data integrity.
Backup, restore, and recovery plan validation. Verification of policies and test strategies as outlined in the VMP.








